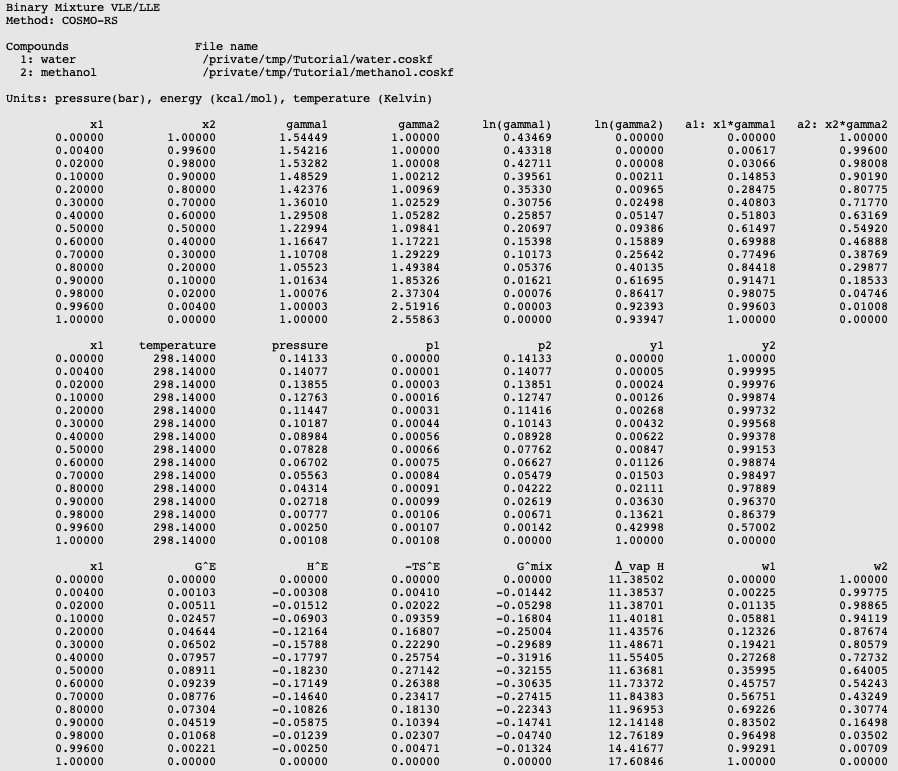
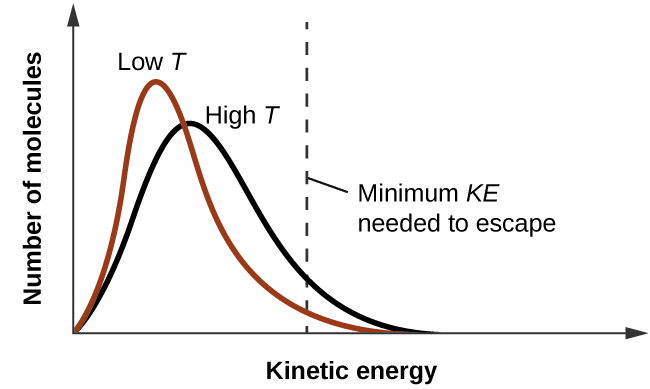
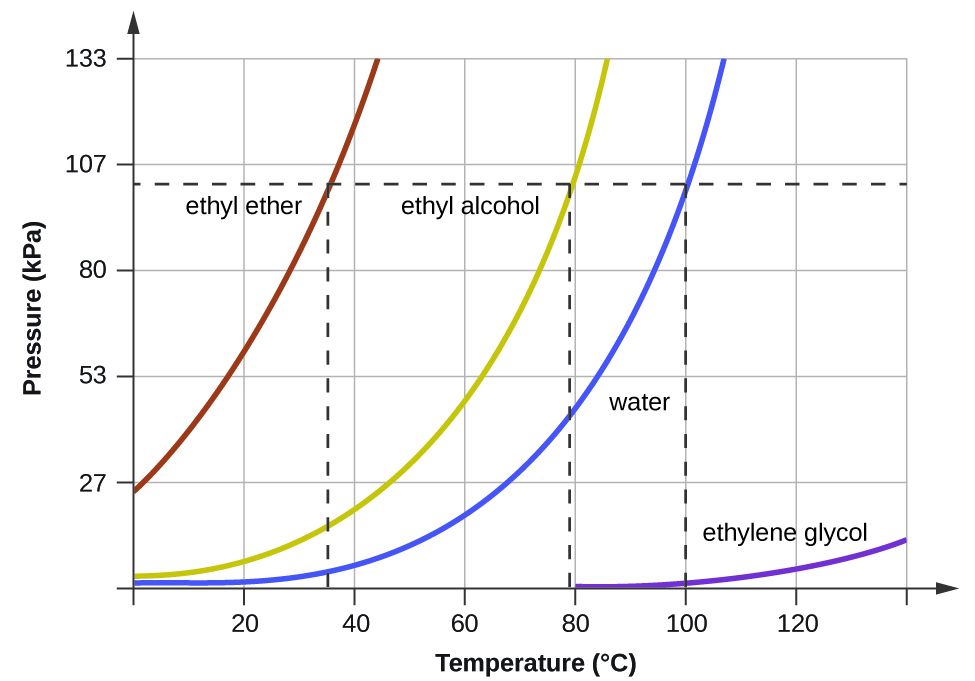
The vapour pressure of ethanol and methanol are 44.0 mm and 88.0 mm Hg, respectively. An ideal solution is formed at the same temperature by mixing 60 g of ethanol with 40g

The vapour pressure of ethanol and methanol ate `44.5 mm Hg` and `88.7 mm Hg`, respectively. - YouTube

The vapour pressures of ethanol and methanol are 44.5 mm and 88.7 mm Hg respectively. An ideal solution is formed at the same temperature by mixing 60 g of ethanol with 40
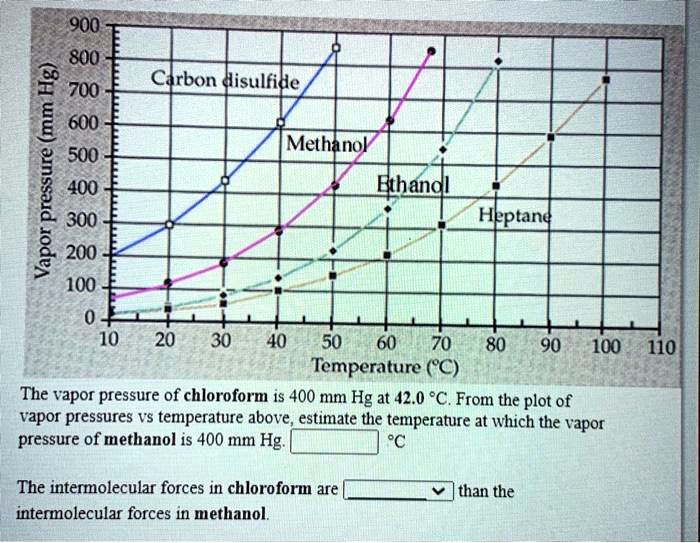
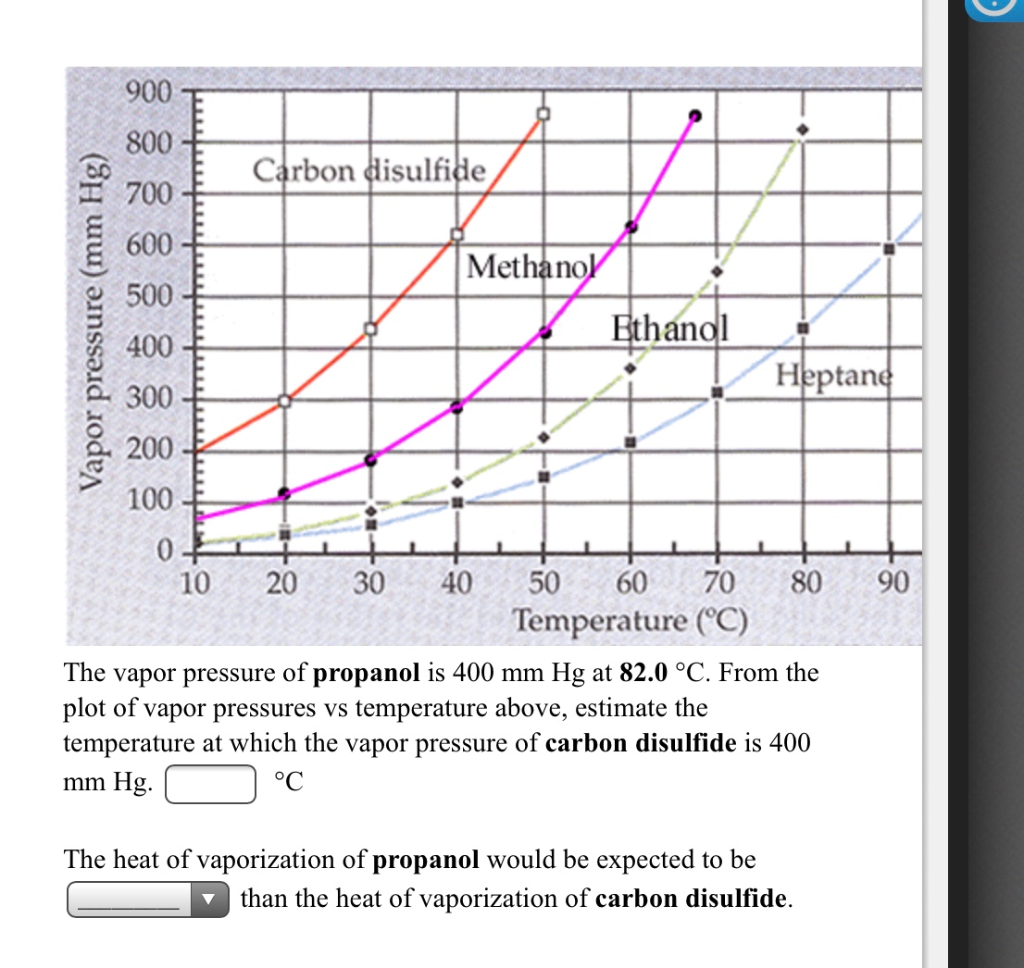
SOLVED: 900 800 2 700 1 600 500 L 400 300 200 100 Ctrbon disulfide Methanol Ethandl HEptand 10 20 30 40 50 60 70 80 Temperature (C) 90 100 110 The

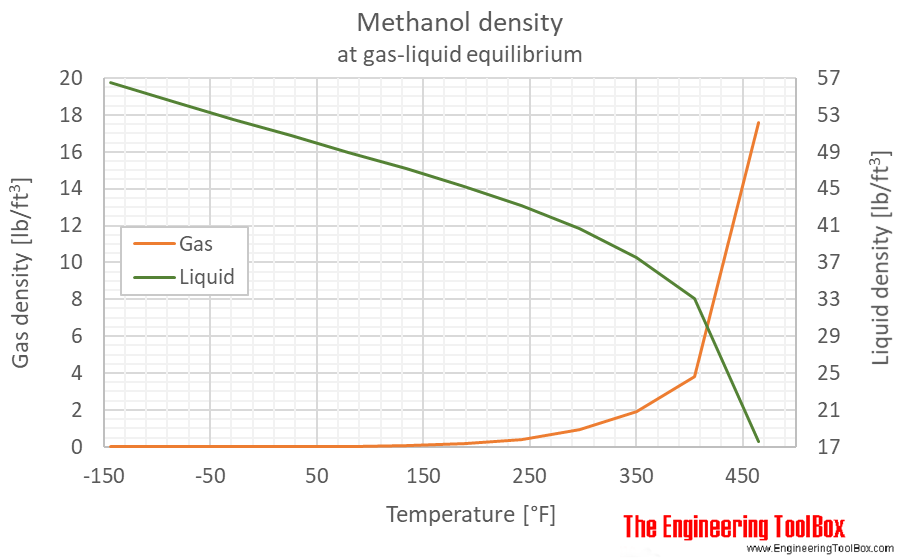
Methanol vapor pressure curve. Markers located at atmospheric pressure... | Download Scientific Diagram

SOLVED:The vapor pressure of methanol, CH3 OH, is 94 torr at 20^∘ C . The vapor pressure of ethanol, is 44 torr at the same temperature (a) Calculate the mole fraction of
ure to answer all parts. The vapor pressure of ethanol (C2H5OH) at 20°C is 44 mmHg, and the vapor - Brainly.com
Solved] 900 800 Carbon disulfide 700 600 Methanol 500 Vapor pressure (mm Hg) Ethanol Heptane 300 200 100 10 20 30 40 50 60 70 80 90 100 110 Temperat... | Course Hero

The vapour pressures of ethanol and methanol are `44.5` and `88.7 mm Hg`, respectively. An ideal... - YouTube