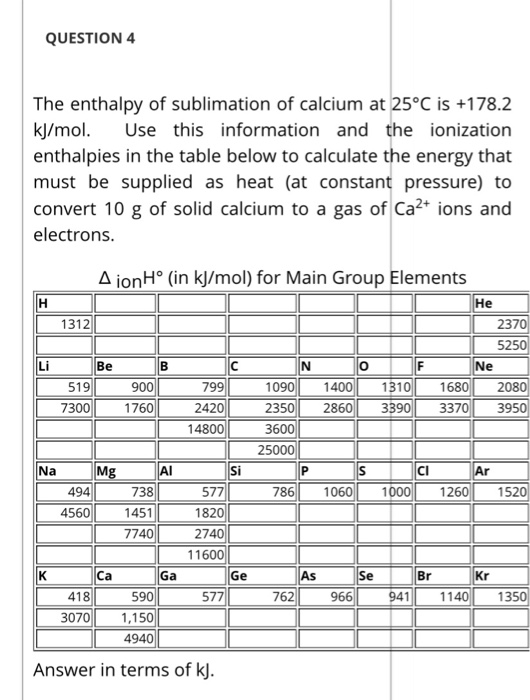
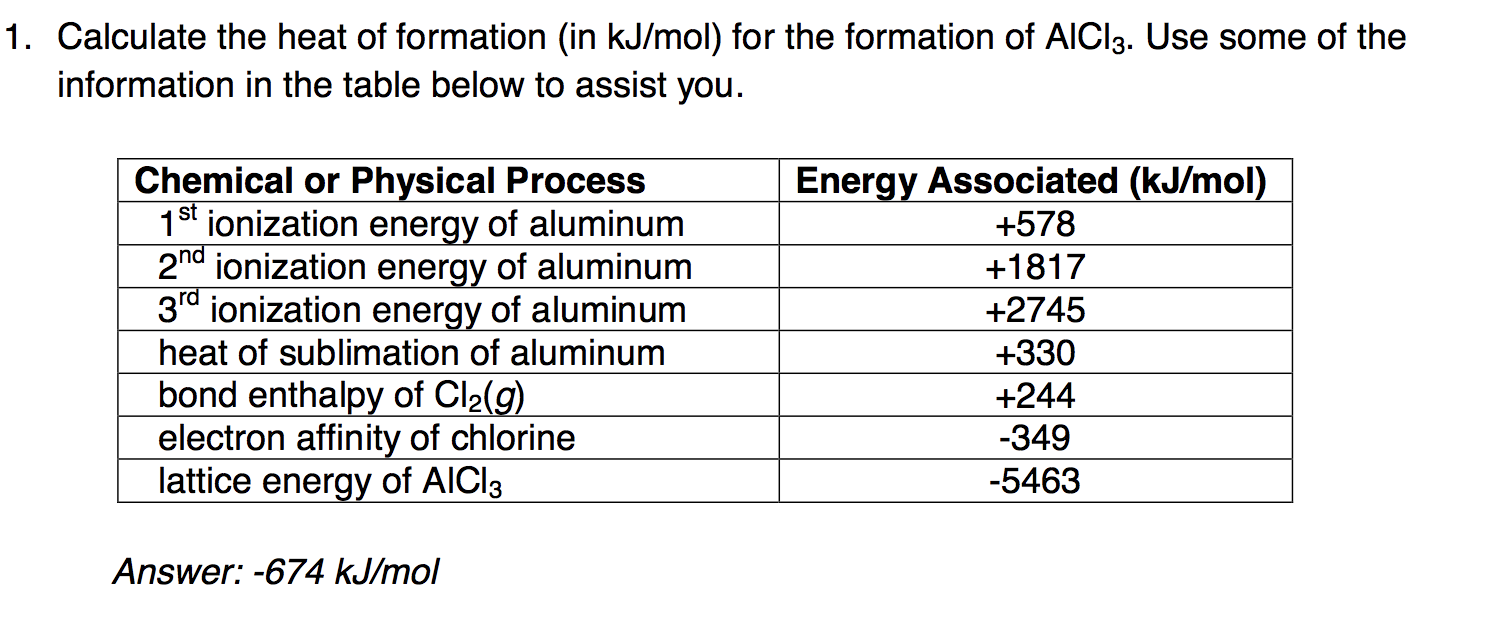
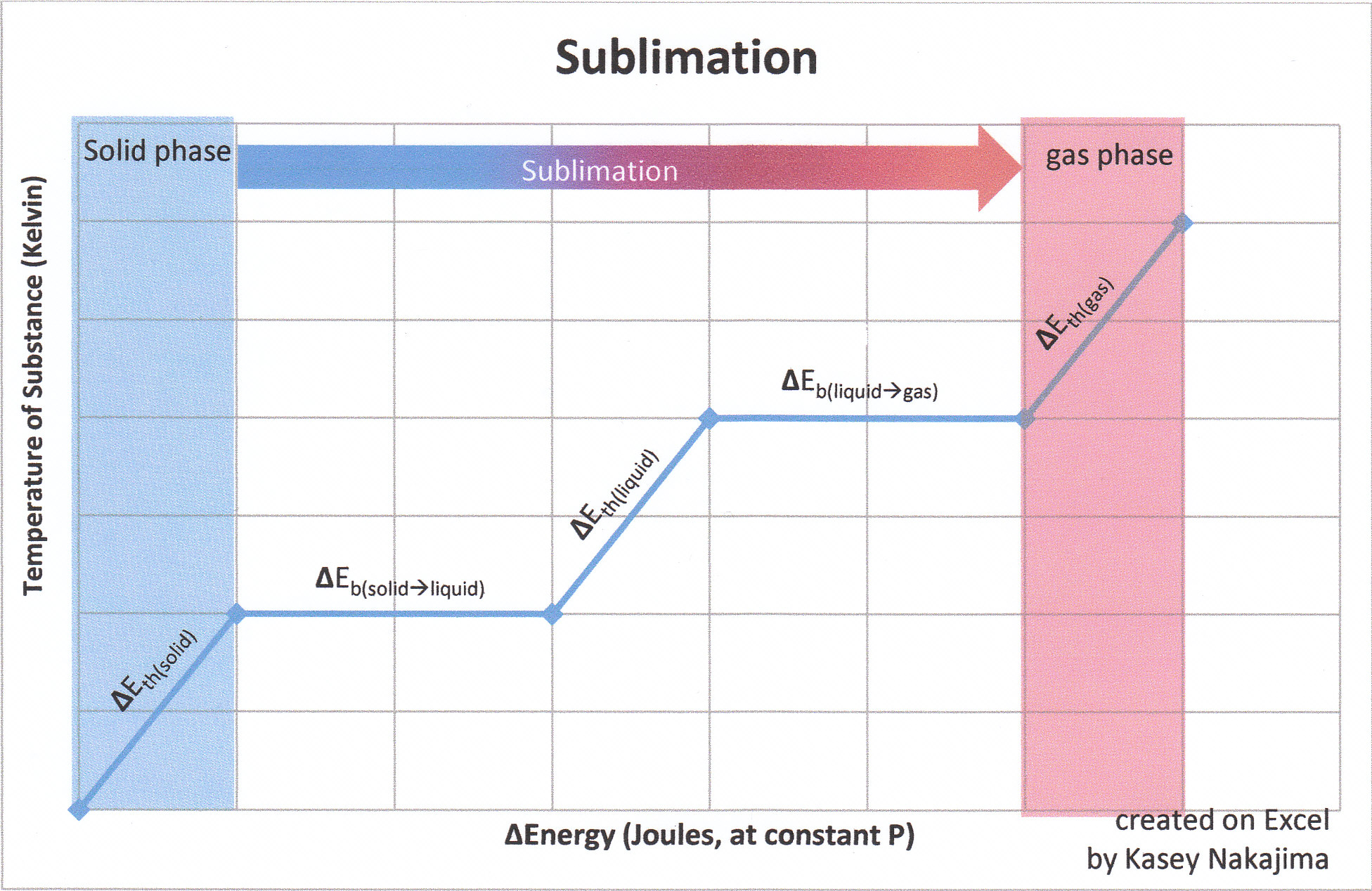
Table 4 from Sublimation enthalpies at 298.15 K using correlation gas chromatography and differential scanning calorimetry measurements | Semantic Scholar

Table 2 from Atom-based thermochemistry: predictions of the sublimation enthalpies of group 12 chalcogenides and the formation enthalpies of their polonides. | Semantic Scholar

Table 3 from Sublimation enthalpies at 298.15 K using correlation gas chromatography and differential scanning calorimetry measurements | Semantic Scholar
![PDF] Sublimation pressure and sublimation enthalpy of H2O ice Ih between 0 and 273.16 K | Semantic Scholar PDF] Sublimation pressure and sublimation enthalpy of H2O ice Ih between 0 and 273.16 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/389cffc31c09d0d5f9a730d417979a3d2d77998b/4-Table1-1.png)
PDF] Sublimation pressure and sublimation enthalpy of H2O ice Ih between 0 and 273.16 K | Semantic Scholar

Table 2 from Enthalpies of Sublimation of Organic and Organometallic Compounds. 1910–2001 | Semantic Scholar

Sublimation Enthalpies of Organic Compounds: A Very Large Database with a Match to Crystal Structure Determinations and a Comparison with Lattice Energies | Crystal Growth & Design
![PDF] Phase Transition Enthalpy Measurements of Organic and Organometallic Compounds. Sublimation, Vaporization and Fusion Enthalpies From 1880 to 2010 | Semantic Scholar PDF] Phase Transition Enthalpy Measurements of Organic and Organometallic Compounds. Sublimation, Vaporization and Fusion Enthalpies From 1880 to 2010 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/930c0b666480121ed889dacb16367b1c0231455d/5-Table1-1.png)
PDF] Phase Transition Enthalpy Measurements of Organic and Organometallic Compounds. Sublimation, Vaporization and Fusion Enthalpies From 1880 to 2010 | Semantic Scholar