Identifying adverse drug event information in clinical notes with distributional semantic representations of context - ScienceDirect

National and institutional trends in adverse events over time: a systematic review and meta-analysis of longitudinal retrospective patient record review studies
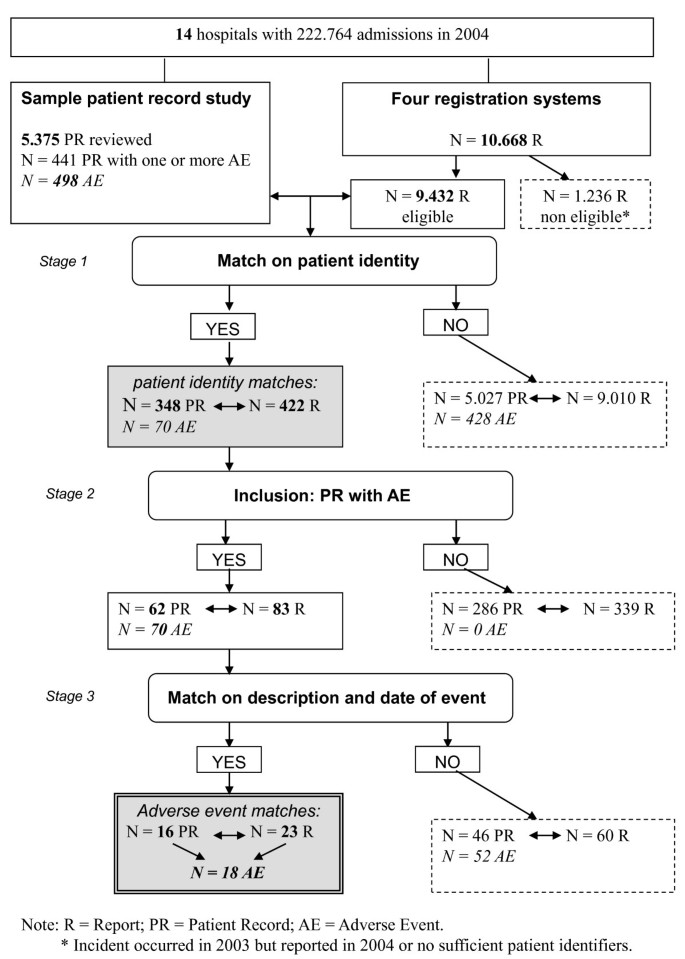
To what extent are adverse events found in patient records reported by patients and healthcare professionals via complaints, claims and incident reports? | BMC Health Services Research | Full Text

Recording Adverse Events Following Joint Arthroplasty: Financial Implications and Validation of an Adverse Event Assessment Form - ScienceDirect

Rates of laboratory adverse events by course in paediatric leukaemia ascertained with automated electronic health record extraction: a retrospective cohort study from the Children's Oncology Group - The Lancet Haematology

Adverse event (AE) reporting algorithm. Timeframe for adverse event... | Download Scientific Diagram
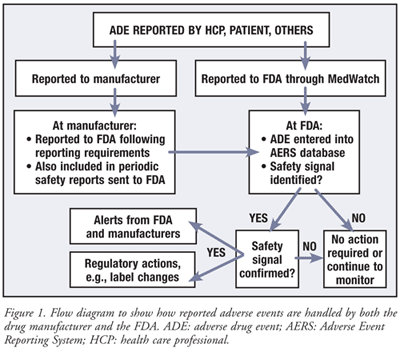
Adverse Event Detection, Processing, and Reporting - Registries for Evaluating Patient Outcomes - NCBI Bookshelf
Documenting, Recording, and Reporting of Adverse Events and Unanticipated Problems Introduction Adverse Events Challenges in Onc

Design of a medical record review study on the incidence and preventability of adverse events requiring a higher level of care in Belgian hospitals – topic of research paper in Clinical medicine.

Adverse event recording failed to reflect potential harms: a review of trial protocols of behavioral, lifestyle and psychological therapy interventions - Journal of Clinical Epidemiology