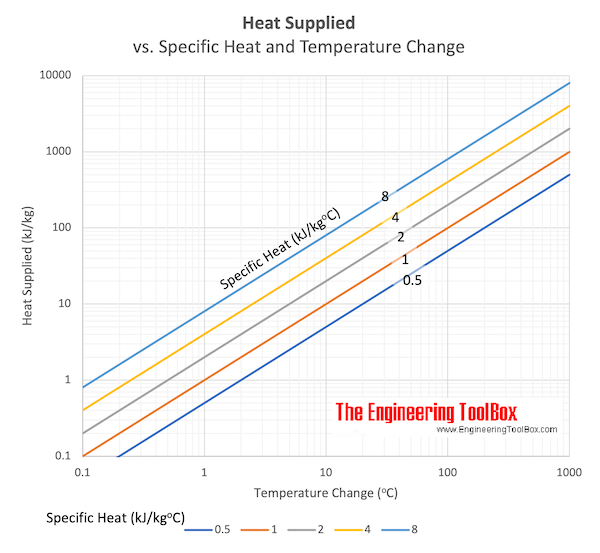
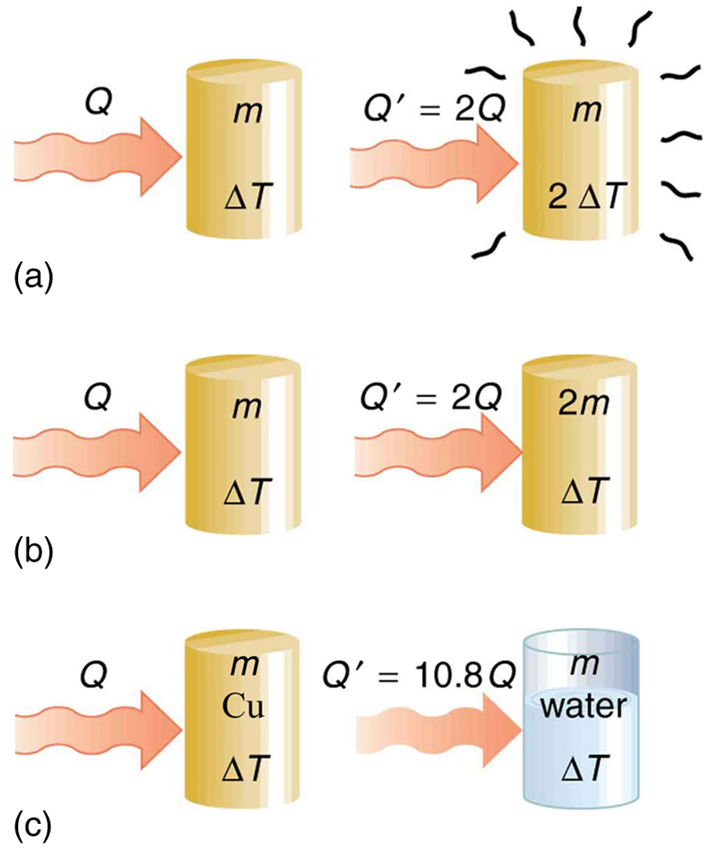
In a container of negligible heat capacity, 200 gm ice at 0^(@)C and 100gm steam at 100^(@)C are added to 200 gm of water that has temperature 55^(@)C. Assume no heat is

Week #1 Quarter 3 (1/3-1/17) (calendar site)(calendar site) Monday, 1/13 Pick Up: Handouts Have out: Temperature and Heat Outline Notes Pick Up: Handouts. - ppt download

Temperature and thermal expansion Specific Heat Capacity Phase changes and Heat Molecular picture of a gas Ideal gas law Kinetic theory of. - ppt download

Question Video: Finding the Specific Heat Capacity of a Substance given the Change in Its Temperature and Internal Energy | Nagwa