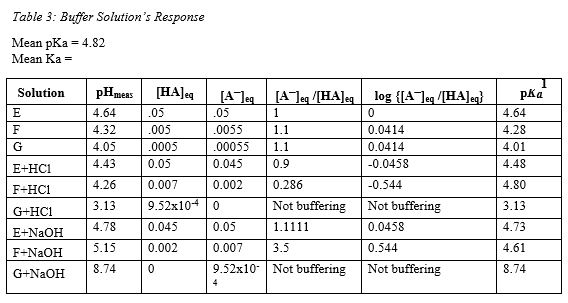
equilibrium - Why do buffers need to be composed of equal amounts of the acid and salt? - Chemistry Stack Exchange
![PDF] Buffers A guide for the preparation and use of buffers in biological systems | Semantic Scholar PDF] Buffers A guide for the preparation and use of buffers in biological systems | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b2a579114d34f9351013ad084db8ea580e8ffd6d/18-Table2-1.png)
PDF] Buffers A guide for the preparation and use of buffers in biological systems | Semantic Scholar
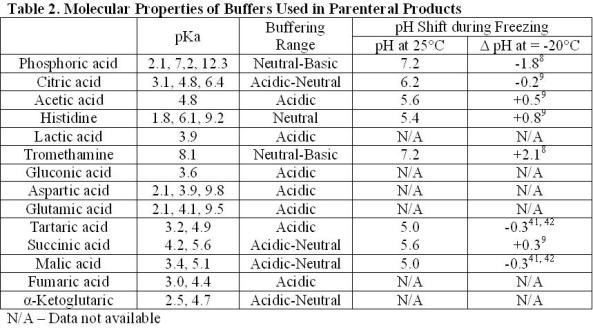
Breaking old habits: Moving away from commonly used buffers in pharmaceuticals - European Pharmaceutical Review
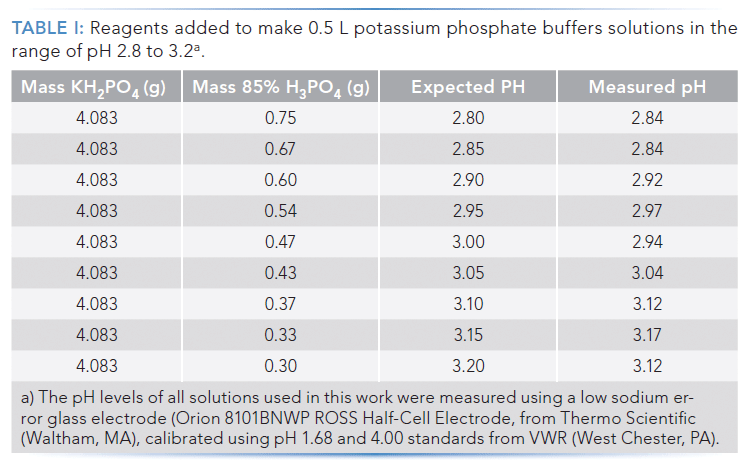
Mobile Phase Buffers in Liquid Chromatography (LC): Effect of Buffer Preparation Method on Retention Repeatability

Table 2 from Values of the standard succinate buffer solution in the 50 mass % ethanol — water solvent | Semantic Scholar

Table 4 from Buffer Standards for the Physiological pH of N-(2-Hydroxyethyl) piperazine-N'-4-butanesulfonic Acid (HEPBS) from 5 to 55 °C | Semantic Scholar
Table of used printing buffer, used buffer concentrations and pH with... | Download Scientific Diagram