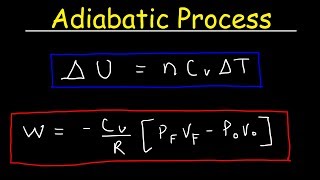
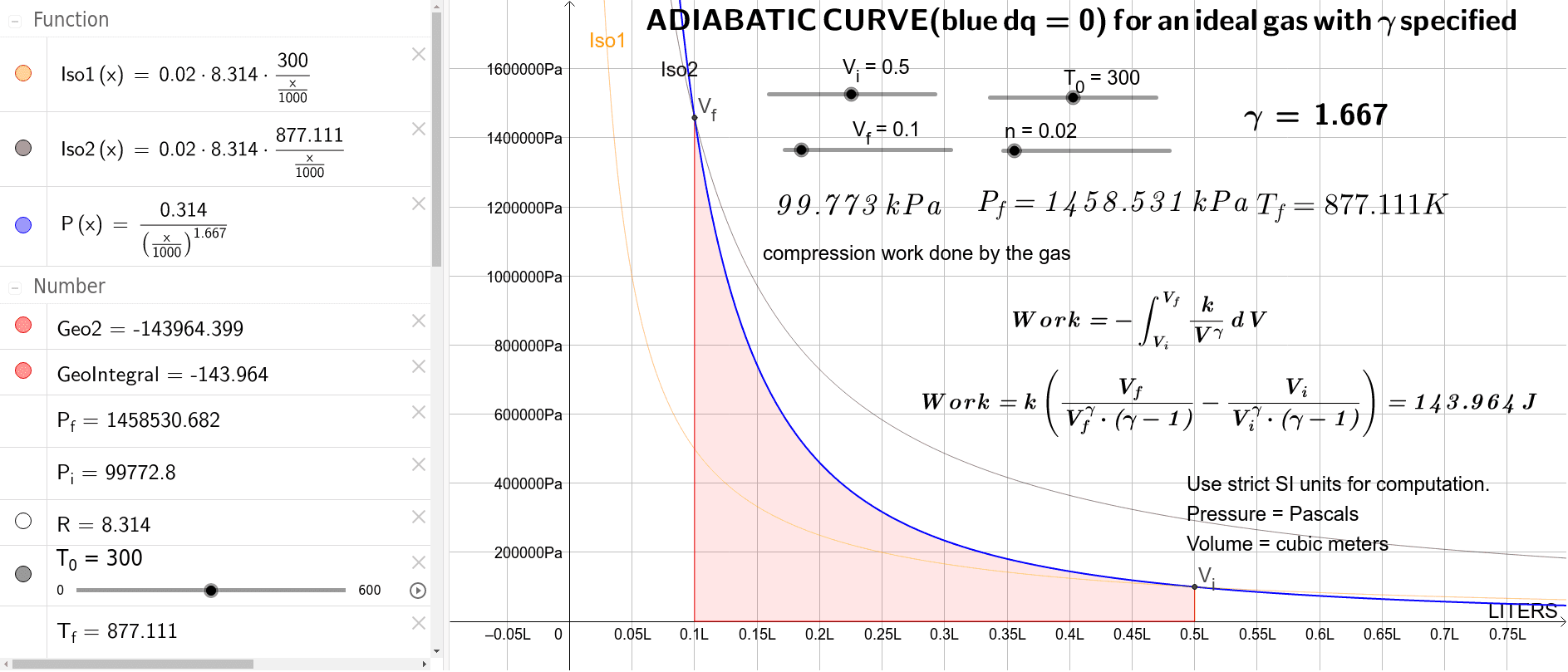
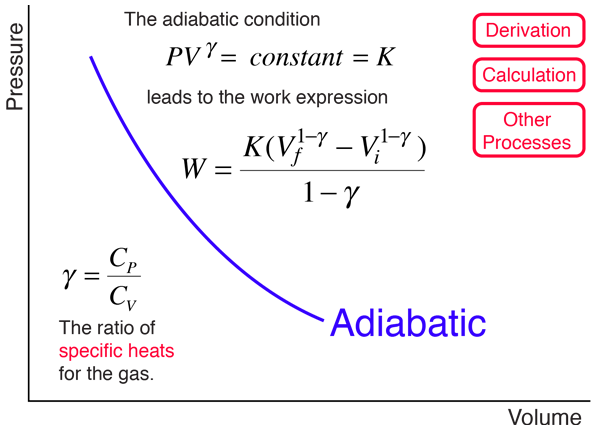
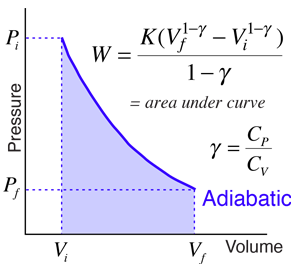
Explain why Boyle's law cannot be used to calculate the volume of a real gas when it is converted from its initial state to final state by an adiabatic expansion.

SOLVED: 12 Calculate the temperature after adiabatic compression of a gas to 10.0 atmospheres pressure from initial conditions of 1 atmosphere and 340 K for air (Y = 1.4) * (2.5 Points) 723 K 657 K 580 K 696 K 618 K
Adiabatic-temperature rise: an awkward calculation made simple: how to estimate a final temperature after a reaction - Document - Gale Academic OneFile

Adiabatic Equation Calculator for Android : Version 1.0 | Equation, Mathematical equations, Calculator