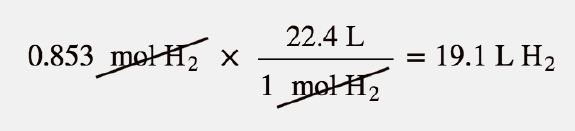Gases & Stoichiometry. Molar Volume 1 mol of gas = 22.4 L molar volume What volume would be occupied by 0.77 moles of helium gas at STP? - ppt download

Gases & Stoichiometry. Molar Volume 1 mol of gas = 22.4 L molar volume What volume would be occupied by 0.77 moles of helium gas at STP? - ppt download

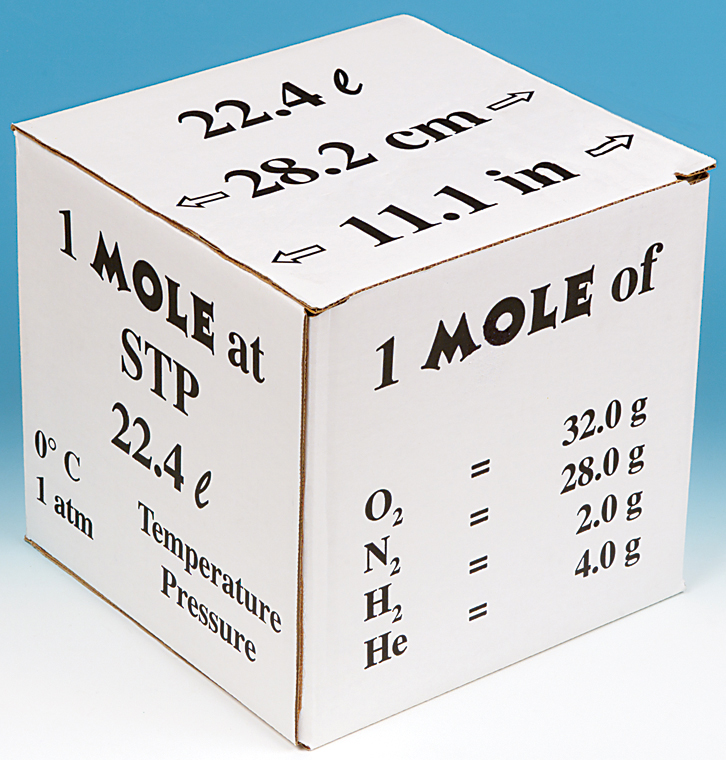
Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP : 1 atmospheric pressure, 0^oC ). Show that it is 22.4 litres .

Molar volume is the volume occupied by 1 mol of any (ideal) gas at standard temperature and pressure (STP : 1 atmospheric pressure, 0^oC ). Show that it is 22.4 litres .

SOLVED:Suppose you have 22.4 L of the following gases at STP: neon, Ne, argon, Ar, and xenon, Xe. a. How many atoms are there in each gas sample? b. What is the